This in-depth and detailed article is provided by Eshani King, a researcher in immunology and health. Part two of this blog can be found here.
It is now widely known that a disproportionate number of COVID-19 related mortalities in the UK have been in non-white, BAME populations [2]. The obese, diabetics, hypertensives and the elderly have also been disproportionately affected. BAME doctors account for 94% of total deaths of doctors but comprise 44% of doctors [1]. The suggestion that these deaths result from deprivation does not fit with the high numbers of deaths of doctors and others in higher socio-economic classes; there must be a different and perhaps more comprehensive explanation.
This review examines the compelling body of evidence strongly implicating varying levels of serum vitamin D levels in the significantly disparate outcomes between different groups of people and between different countries. It explores the extent of vitamin D deficiency, highlighting countries and categories of people most likely to be deficient. It demonstrates that the UK suffers from exceptionally high levels of vitamin D deficiency with serum levels averaging only 20ng/ml, half the optimal level; It examines the strong body of existing evidence connecting vitamin D deficiency to increased respiratory tract infections highlighting the central epigenetic role of vitamin D in immune system responses during a respiratory tract infection with SARS-CoV-2; It reviews research flagging correlations between COVID-19 outcomes and vitamin D deficiency and studies providing the first direct evidence linking low vitamin D status with worse outcomes from COVID-19. Cautions regarding future trial designs and lack of evidence for toxicity concerns are both discussed. Current UK guidelines recommend 400 IU of vitamin D per day whereas 6,000-10,000 IU per day is required to maintain blood levels of 40ng/ml, widely accepted as being the level required to support immunity, optimal health, and reduction in all-cause mortality. The financial cost of vitamin D deficiency in the UK has been estimated at around £20 billion per annum.
2. Introduction to Vitamin D
Vitamin D3 (cholecalciferol), a fat-soluble vitamin, is made by the action of UVB sunlight on 7-dehydrocholesterol in the skin and converted in the liver to 25(OH)D or 25-hydroxyvitamin D, occasionally referred to as calcidiol. This is the form that circulates in the blood at detectable levels and measured in order to determine serum vitamin D status. The kidneys carry out a second hydroxylation by the enzyme 1-α-hydroxylase to convert 25(OH)D to 1,25 (OH)D or 1,25-di-hydroxyvitamin D, also referred to as calcitriol or active vitamin D [17]. This second hydroxylation to make the active form, calcitriol, has also been found to occur in many other cells including those in the respiratory tract epithelia and cells involved in the immune response. Calcitriol is 1000 times as potent as the inactive circulating 25(OH)D form but there is around 1,000 times less of it. Please note that, except where otherwise stated, the term vitamin D has been used interchangeably throughout to refer to any of the three forms.
The calcitriol form of vitamin D functions as a hormone in every cell of the body, all of which have vitamin D receptors (VDR). VDRs are nuclear transcription factors that epigenetically control the expression of up to 2,000 different genes including almost 300 genes just in white blood cells involved in the immune response [79]. There is no doubt that Vitamin D is of critical importance to many aspects of health. In addition to its long-known role in bone health, it also plays an important and central role in immune-enhancement and immune-modulation.
A note regarding units: ng/ml has been used throughout for serum levels of vitamin D. To convert ng/ml of 25(OH)D to nmol/l the conversion factor is 2.5.
3. How Widespread is Vitamin D Deficiency?
3.1 Vitamin D Deficiency in BAME Populations
It is generally acknowledged that BAME populations in the west are vitamin D deficient because the screening effect of melanin pigment in darker skin means far stronger sunlight is required to make vitamin D from sun exposure [17]. Although a serum level of 20ng/ml is generally accepted as the level necessary to prevent deficiency (and resultant conditions such as rickets, osteoporosis and osteomalacia), it is thought by many experts in this field that a far higher level of 40-60ng/ml or more is required to achieve optimum immune system and cancer fighting capability, with 30 ng/ml being “sufficient” but not optimal [13,52,74]. 40-60ng/ml is also the level required to achieve normalised parathyroid function [3-10] and also the level maintained by indigenous peoples such as the Maasai despite their very dark skin [61,62]
Darker skins cannot make enough vitamin D at latitudes above the 37th parallel even in summer and even white skins cannot make vitamin D in the winter months when the UVB light is much weaker [4-6,17]. Furthermore, studies have found that men generally have lower vitamin D levels than women [70].
The following is a table produced by Grassroots Health, USA showing rates of deficiency and sufficiency in different ethnic groups in the US [11]. The differences are very striking; Only 6% of black ethnic groups have optimal vitamin D levels of 40ng/ml but surprisingly, only between 15-20% of white skinned individuals attain this level. The most dramatic difference is that whilst only around 15% of the white population is deficient, meaning they have levels below 20ng/ml, between 55-60% of the black ethnic population is deficient. The vitamin D status of Asians, Hispanics and others lie in between these extremes.
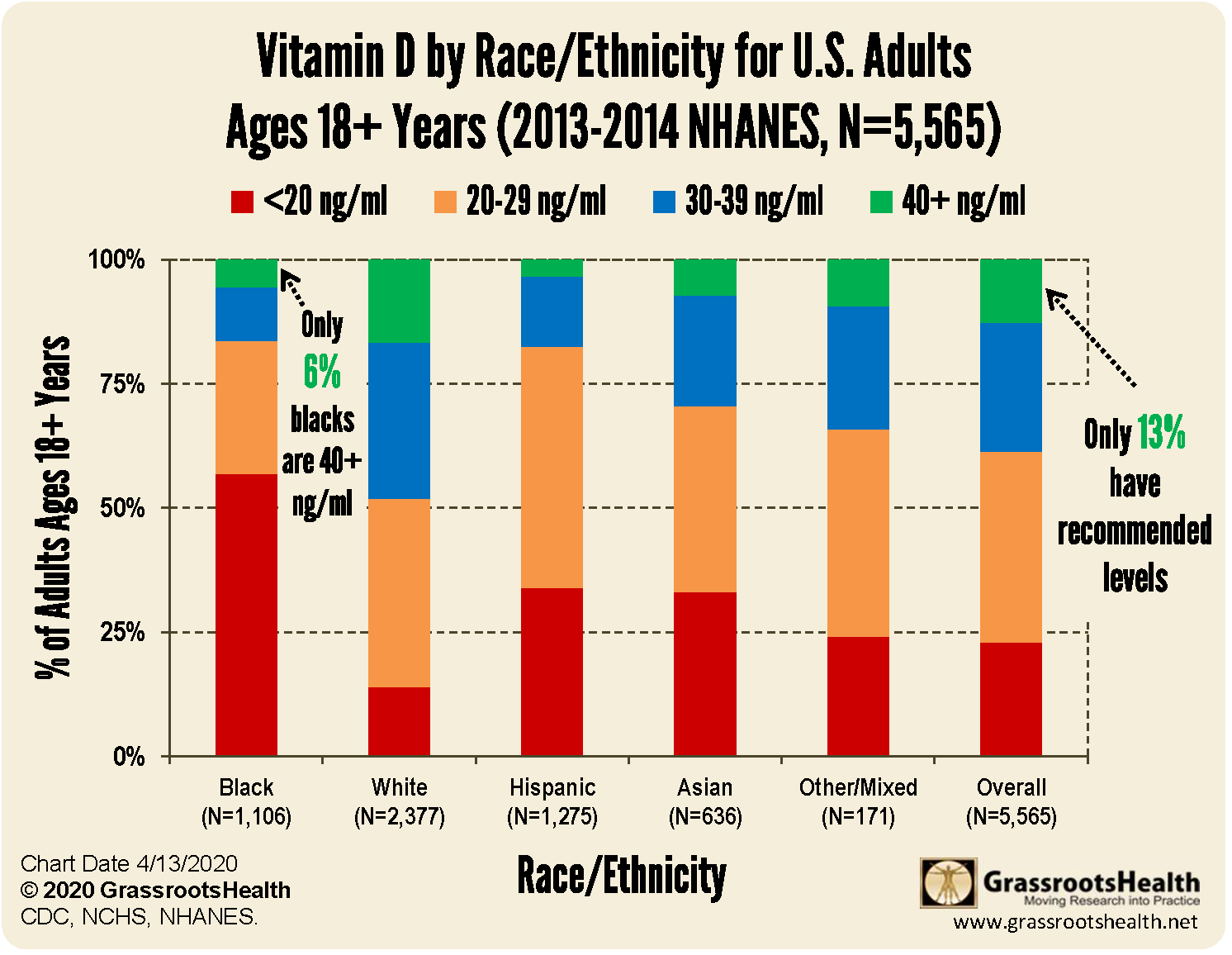
This demonstrates that vitamin D deficiency is not just an issue affecting black/dark skinned individuals but is far more widespread, with many who are white skinned also suffering from vitamin D insufficiency and less that 20% of them achieving optimal serum levels of at least 40ng/ml.
3.2 Vitamin D Deficiency in the UK and Europe
The above statistics, as noted, are from the US, and it may be expected that the situation is better in the UK. Unfortunately, it appears to be surprisingly worse.
The following is a comparison of deficiency rates for different European countries rounded to the nearest whole number. Although the data is taken from different studies collated in a 2019 study by Paul Lips using different population sets [12], it can nevertheless provide some useful comparisons. Where there are two studies relating to one country, the average values have been used (as the two figures were generally close), rather than a weighted average. No statistics were provided for Spain.
Average Vitamin D levels and rates of deficiency by country
| Country | Average ng/ml | % <10ng/ml | % <20ng/ml |
| Norway | 27 | 0 | 17 |
| Sweden | 29 | 0 | 16 |
| Iceland | 23 | 4 | 34 |
| Denmark | 26 | 0 | 24 |
| Germany | 21 | 5 | 50 |
| UK | 19 | 15 | 56 |
| N Ireland | unavailable | 17 | 66 |
| Ireland | 23 | 6 | 45 |
| Belgium | 20 | 8 | 51 |
| Netherlands | 27 | 4 | 31 |
| France | 24 | 6 | 35 |
| Greece | 20 | 2 | 51 |
| Italy | 24 | unavailable | 52 |
The table shows a startling level of vitamin D deficiency in the UK and N Ireland; 56.4% and 66.2% of the population have levels below 20ng/ml; similar to that seen in the American black population in the table above. This makes the UK the most deficient country in the table with average blood levels of vitamin D of 19ng/ml, followed closely by Belgium with 19.7ng/ml. However, no other country comes near the UK for severe deficiency; the percentage of the population with levels below 10 ng/ml is over 15%, with Belgium again in second place with 7.3%.
In addition to the disproportionate number of COVID-19 related deaths in the BAME communities in the UK, it is of note that the Somali population of Sweden has also suffered a disproportionate number of deaths: despite comprising less than 1% of the population, press reports as at 11 May state that they accounted for 18% of COVID-19 related deaths countrywide and 40% of Stockholm’s COVID-19 related deaths.
Generally, however, vitamin D status in Scandinavian countries is, given their latitude, surprisingly better than southern European countries. This is thought to be the result of widespread awareness of the issue, food fortification having been in place for many years, and a far higher consumption of vitamin D containing oily fish and fats [82]. It is interesting that as a result of a review by the independent Swedish Council on Health Technology Assessments of 16,000 studies through May 2013, the Swedish were advised to consume plentiful quantities of animal fats and cream and to eliminate/reduce sugar and carbohydrates as a strategy to prevent diabetes, cardiovascular disease, and obesity. Meanwhile, in contrast, nutritional advice in the UK has been geared strongly towards a low animal fat diet.
3.3 Vitamin D Deficiency Elsewhere
Even in hot, sunny countries, vitamin D deficiency can still be rife because of full clothing and deliberate sun avoidance for cultural reasons, e.g. in some parts of the Middle East and in certain socio-economic groups in India. Rural and slum dwellers possibly achieve higher levels of sun exposure and therefore better vitamin D levels. The percentage of the population with <20ng/ml in India and the Middle East is remarkably high, 75%, and 90%, respectively, compared to the USA, Australia, and Canada: 36%, 31% and 61% [12,13].
3.4 What Other Categories of People Might be Vitamin D Deficient?
What is perhaps not generally known is that the obese, another high-risk group, are also highly likely to be highly vitamin D deficient as they require 2-5 times (or even more) vitamin D than others, dependent on the level of obesity [4,9,17,52,78]. Unless they are taking exceptionally high levels of supplements they are highly likely to be even more deficient than BAME categories. The fat-soluble vitamin is thought to become trapped in fat cells although the exact biochemistry of this is uncertain. The elderly is another category highly likely to be vitamin D deficient unless they supplement; older skin gradually loses the ability (by up to 75%) to convert 7-dehydrocholesterol to vitamin D [4,9,17].
It is also of note that virtually all of the other remaining categories that have an increased risk from COVID-19 are also likely to have a high level of vitamin D deficiency. Other health conditions well documented in a vast body of medical literature, including interventional studies, shown to be caused by (and not merely associated with) lack of optimal vitamin D include:
- asthma, allergic rhinitis, and other respiratory diseases[30-31]
- autoimmune diseases[17,22,74,75]
- high blood pressure [14-17]
- dermatological conditions [3,9]
- heart disease [17,55,89]
- bone loss [3,12,13,17]
- fatigue (often misdiagnosed as fibromyalgia) [17]
- hair loss
- bone and back pain [3,9,12]
- muscle pain [3,9]
- depression [3,9]
- infertility [3,9,12]
- obstructive sleep apnoea
- acute respiratory infection [36-52]
- nasal polyps
- Otis media
- diabetes [13,18-21]
- chronic rhinosinusitis
- cancer (including breast, melanoma, ovarian, prostrate, head and neck, Hodgkin’s, and non-Hodgkin’s lymphoma) [23-28]
- Alzheimer’s
Unconverted vitamin D, rather than the 25 (OH)D form has been shown to stall the progression of prostate cancer at high concentrations [28].
Although there may also be other factors involved in the above conditions, such as various nutrient deficiencies and the metabolism of excessive quantities of glucose, vitamin D deficiency is a major contributory factor in the genesis of these conditions. Although high serum vitamin D levels could have prevented their onset, subsequent supplementation may or may not be effective in alleviating a condition once it has become established, depending on the nature of the particular conditions.
Despite this, there is still every expectation that increasing the serum levels of vitamin D for people with these conditions, all of whom are highly likely to be vitamin D deficient, can help combat viral infection as this involves different biological mechanisms as illustrated in Section 5 below.
It is of interest that some pharmaceutical drugs including some antihypertensives, antibiotics, antiepileptics and anti-inflammatory agents used to treat some of these conditions can reduce serum vitamin D, exacerbating existing vitamin D deficiency [67]
4. Connection Between Vitamin D Deficiency and Respiratory Tract Infections [36-52]
Multiple cross-sectional studies and interventional studies have associated lower vitamin D levels with a statistically significant increase in rates of infection with respiratory viruses including influenza [42-52].
A recent paper, William B. Grant et al “Evidence that Vitamin D Supplementation Could Reduce Risk of Influenza and COVID-19 Infections and Deaths” April 2020: https://www.grassrootshealth.net/wp-content/uploads/2020/04/Grant-GRH-Covid-paper-2020.pdf discusses the sizeable body of compelling literature evidencing the role of vitamin D in preventing respiratory illnesses, and also suggests possible reasons why no association was found in others.
In addition, Dr Rachel Neal who is currently leading the world’s second largest trial of high dose vitamin D supplementation, reported that a study of 78,000 patients last year found that patients with low levels of vitamin D were twice as likely to develop acute respiratory infections and be sicker for longer that patients with high vitamin D levels [81].
In another study described by Dr Holick, it was demonstrated that supplementing one group of postmenopausal women with 2,000 IU of vitamin D resulted in a 90% reduction in upper respiratory tract infections compared with a group receiving only 400 IU [73p112]. Note that 400 IU is the level currently recommended by Public Health England. In another study, vitamin D blood levels of at least 38ng/ml were associated with a two-fold reduction in the risk of developing acute respiratory tract infections [52]. In contrast, as already noted, the average serum level for the UK is only 19ng/ml.
Although the 1918 Spanish flu is not examined further here it may be of interest that the sunnier US states such as Texas were observed to have had lower death rates than the Northeast.
The seasonality of flu very probably has little to do with the premise that viruses degrade in the heat (which needs far higher temperatures than our UK summers) but more to do with the fact that we rely on sunlight for a high proportion of our vitamin D requirements. Any reserves of vitamin D made in the summer months, meagre as they are, are depleted from September onwards so that by December/January, serum levels become perilously deficient in many.
5. The Critical Role of Vitamin D in the Immunological Response to a COVID-19 Infection
5.1 Vitamin D in the Innate and Adaptive Immune Response
The following discussion is limited to the known biological role of vitamin D in the type of immune responses seen against a SARS type respiratory infection and is by necessity a simplified account, ignoring the involvement of many other immune cells and chemical signals within multiple complex, interactive, cross regulating systems of immune responses.
VDR’s (vitamin D receptors) are abundant in cells involved in the immune response, i.e. macrophages, neutrophils, T lymphocytes (Th1 and Th2) and many others [22,67]. Vitamin D clearly has an important role to play in these cells. It is noteworthy that apart from the kidneys, other locations where the enzyme, 1-α- hydroxylase, (which converts vitamin D into its active form), occurs, are the lung epithelial cells, vascular epithelia, gut epithelia and numerous cells involved in the immune function[22,67].
On detecting a foreign pathogen via toll-like receptors (TLR’s), the transcription of the immune system’s antivirals, human cathelicidin and defensins (which also have fungicidal and bactericidal properties), is rapidly increased in monocytes, neutrophils and in other immune cells of the innate immune system in lung, epithelia, intestine and other surfaces which form an interface with the external environment. This forms part of the first line of defence against invading pathogens. This upregulation in antiviral production has been shown to be highly dependent on sufficient vitamin D [32-38]. In a particularly interesting study, it was observed by researchers that sera from African Americans who are known to have low blood levels of vitamin D was inefficient in inducing production of cathelicidin. When the researchers supplemented the sera with 25(OH)D, cathelicidin levels increased to the same levels seen in white Americans [ 38].
Vitamin D also increases the phagocytic killing ability of immune cells such as natural killer cells, macrophages and neutrophils and reinforces the physical barriers of epithelial cells by maintaining tight junctions [22]. These first line defences of the innate immune system can be highly effective in limiting or preventing progression of infection at an early stage, resulting in no symptoms (asymptomatic) or only mild symptoms: it is clear that this stage is highly dependent on vitamin D status. As discussed at Section 5.3, prior infection with pathogens expressing cross-reacting antigens can also result in asymptomatic or mild cases.
In critical cases of COVID-19, many inflammatory cytokines become significantly elevated. It has been confirmed that interferon-γ, and both Th17 and TH1 cells which highly express inflammatory cytokines, are all involved in acute lung injury during the late phase of SARS infection [84,85,86]. Vitamin D has been shown to inhibit Th17 pro-inflammatory cytokines [87] and Th1 cytokines ( IL-2 and IL-17, IL-6, IL-1β and interferon-γ, etc) by suppressing production of angiotensin II (see further below), but increase expression of anti-inflammatory Th2 cytokines by macrophages. Induction of T regulatory cells by the action of vitamin D further suppresses the inflammatory response [22,67]. Vitamin D deficiency therefore unbalances the immune response by allowing the unchecked dominance of the pro-inflammatory cytokines to create the type of uncontrolled cytokine storms which directly impact the respiratory system, resulting in damage and vascular permeability, as seen with some COVID-19 deaths. In some, ARDS (acute respiratory distress syndrome) can be a potentially disastrous outcome [39-41].
Vitamin D upregulates expression of VDRs and ACE2 (angiotensin converting enzyme 2) which has been shown to be protective against acute lung injury [40]. As part of the RAS (renin-angiotensin) system, ACE2 is critical in suppression of the angiotensin II induced inflammatory pathways. It does this by converting angiotensin II to anti-inflammatory angiotensin A(1-7). Meanwhile, in a double manoeuvre, the upregulated VDR blocks transcription of renin, an enzyme required to produce angiotensin 1, an intermediary of angiotensin II, thus depressing levels of angiotensin II further [40,41]. However, as SARS viruses also dock at ACE2 receptor sites, the up regulation of ACE2 (which is dependent on vitamin D as noted above,) is critical in preventing a cascade of inflammation. If SARS blocks sufficient ACE2 and upregulation is insufficient, inflammation cannot be effectively controlled. Either insufficiency of vitamin D, a very high viral count, or an immune system which has been previously mis-programmed towards a skewed immune response, away from the anti-inflammatory and towards the inflammatory state (the factors precipitating mis-programming being outside the scope of this analysis), can all potentially result in a SARS type virus gaining the upper hand, before or while antibodies are being produced.
In addition, apparently uniquely in COVID-19 cases, the virus has an effect on red blood cells, precipitating breakdown of haemoglobin, thereby preventing red blood cells from carrying oxygen. This leads to dangerously low levels of oxygen, or hypoxia, which cannot be resolved by ventilators, and the formation of blood clots which further impede airways. It is of interest here that a number of studies demonstrate the anticoagulant effects of vitamin D [72 and references therein] and link lack of vitamin D to increased blood clotting and DVT (deep vein thrombosis). Vitamin D induced suppression of inflammatory cytokines include those cytokines involved in clot formation e.g. IL-22 which upregulates fibrinogen formation [87]. Therapeutic use of vitamin D to dissolve blood clots in DVT patients has been proposed [72], although if a patient has optimal levels of vitamin D to begin with, given the evidence so far presented in this review, they may never get as far as this dangerous stage.
Given the descriptions in this section, the strength of response of the innate immune system’s first line defence system, which is highly dependent on vitamin D status, is potentially of far greater importance that in a non-SARS, non-COVID-19 respiratory infection as that would determine whether the infection proceeds any further. Whilst the innate response happens within hours, the adaptive immune response, the arm of the immune system that produces antibodies, is slower to respond. It is well established that the antibody response can take 5-7 days to come into play.
In the adaptive immune response, vitamin D acts as an immune modulator; antigen presenting cells and T and B lymphocytes involved in antibody production against pathogens are prevented from being over expressed by the presence of sufficient levels of vitamin D, thus preventing the type of hyper-stimulation which can trigger autoimmunity [22,74,75]. Lack of sufficient vitamin D has been implicated in many auto-immune disorders such as lupus, Sjogren’s and others [17].
Vitamin D also upregulates the expression of glutathione, the human body’s antioxidant which also acts as an antiviral and reduces the inflammatory response [83].
Vitamin D is therefore critical for a rapid, effective but balanced and controlled immune response which does not result in overactivity and autoimmunity.
5.2 Gender Differences: Major genes coding for innate immunity are located on the X-chromosome. Women therefore have two copies and men, one. The interaction of these genes together with their generally higher vitamin D status as well as hormonal factors are thought to result in women being less adversely affected by COVID-19 and by all infections generally than men [68-70].
5.3 Asymptomatic and Mild Cases: It has already been demonstrated that a strong innate response underpinned by optimal vitamin D status can be successful in halting progression of infection. This is what very probably happens in many of these cases. There is also another quite significant category of people that are likely to already have some immunity to SARS-CoV-2 despite never having encountered it before. Recent research, detailed below, testing blood samples including blood from 2015-2018 blood banks has demonstrated that between 34%, and 50% of people have T-helper cells that cross-react with SARS-CoV-2 antigens from the spike protein and also with non-spike proteins. These are also present in people who have actually had COVID-19 but in those that have not, the authors presume they were acquired through prior infections with one of the 4 common cold coronaviruses. However, no follow up studies have been carried out to demonstrate that these people will have mild, less severe or no symptoms when they encounter SARS-CoV-2.
Immunologist Andreas Thiel’s research at Charite University Hospital, Berlin: https://www.medrxiv.org/content/10.1101/2020.04.17.20061440v1
Immunologists Shane Crotty and Allessandro Sette’s research at La Jolla Institute for Immunology: https://www.cell.com/cell/fulltext/S0092-8674(20)30610-3
Helper T-cells such as those in these studies take messages to antibody producing B cells, stimulating them to upregulate production of antibodies. They also stimulate killer T-cells to kill pathogens and send signals to memory T-cells which are thought to retain long lasting immune memory. The correct balanced expression of such T-cells is also controlled by vitamin D [75].
5.4 Children have less developed and less specialised adaptive immunity and are more dependent on their innate immune response. The maturing immune system is also primed towards an anti-inflammatory response[1]. In addition, they tend to have greater sun exposure and therefore higher vitamin D levels. They also have greater exposure to dirt and a higher level of interaction with micro-organisms in the general environment, all of which re-enforce and strengthen the innate immune response and play a critical role in maturation of the developing adaptive immune system. The stronger innate and anti-inflammatory response might be reasons why children are far less affected and are mostly asymptomatic.
It should be noted that despite becoming immune following a COVID-19 infection, it is possible that young children might not produce much in the way of specific antibodies which can be detectible in a COVID-19 antibody test. Antibodies in children can be broad spectrum rather than highly specific, capable of dealing with a more varied range of antigens [76]. Immune memory can also reside in T-cells as noted above, and further research is required in order to establish exactly where immune memory resides in young children.
6. New Evidence linking COVID-19 Related Deaths to Vitamin D Deficiency
6.1 Connections and Correlation Studies
A recent study of 20 European countries by Queen Elizabeth Hospital Foundation Trust found a correlation between level of vitamin D deficiency and COVID-19 related deaths and cases. https://link.springer.com/article/10.1007/s40520-020-01570-8
The data for this has been taken from the same source as the table in section 3.2. These are interesting findings, although they may be slightly limited by the fact that countries are at different stages in the infection curve and the criteria used to classify a death as being COVID-19 related may differ in different countries. More significantly, the death rates reported will be dependent on the level of testing in each country until antibody testing establishes the true extent of infection and therefore the true death rate per million of population. However, those factors are true for all country comparisons and it will be very interesting to see if the correlation still holds when more directly comparable data becomes available for all the countries in the study.
It is, however, of note that the UK and Belgium, the very two countries that have the highest levels of vitamin D deficiency (Section 3.2 ) are also those with very high numbers of COVID-19 related deaths per million of population.
A recently published pre-print of research by Northwestern University analysing data from 10 countries found a strong correlation between low vitamin D levels and cytokine storms in COVID-19 patients as well as a correlation between vitamin D deficiency and mortality: https://www.medrxiv.org/content/10.1101/2020.04.08.20058578v4, with commentary by the researchers at: https://www.sciencedaily.com/releases/2020/05/200507121353.htm
Recent Irish research published by Trinity College, Dublin, the TILDA study, https://tilda.tcd.ie/publications/reports/pdf/Report_Covid19VitaminD.pdf
, also supports the premise that vitamin D deficiency is widespread and is likely to be a major factor in poor outcomes from COVID-19.
Another point of interest is that UVB light required to make vitamin D is blocked very effectively by air pollution. Northern Italy and Wuhan where COVID-19 related death rates were much higher than elsewhere are both known to suffer from particularly poor air quality. Whilst air pollution is thought to be a factor in the very high death rates in these two areas, lack of vitamin D may have compounded this effect.
It is also of note that sunny countries where sun avoidance is not common, for example, most African countries, Australia, Thailand, Sri Lanka, Hong Kong, South Korea, the Caribbean, and many countries around the equator appear to have escaped with very few deaths, Brazil being a notable exception.
6.2 Direct Evidence for Vitamin D Deficiency in COVID-19 Related Deaths
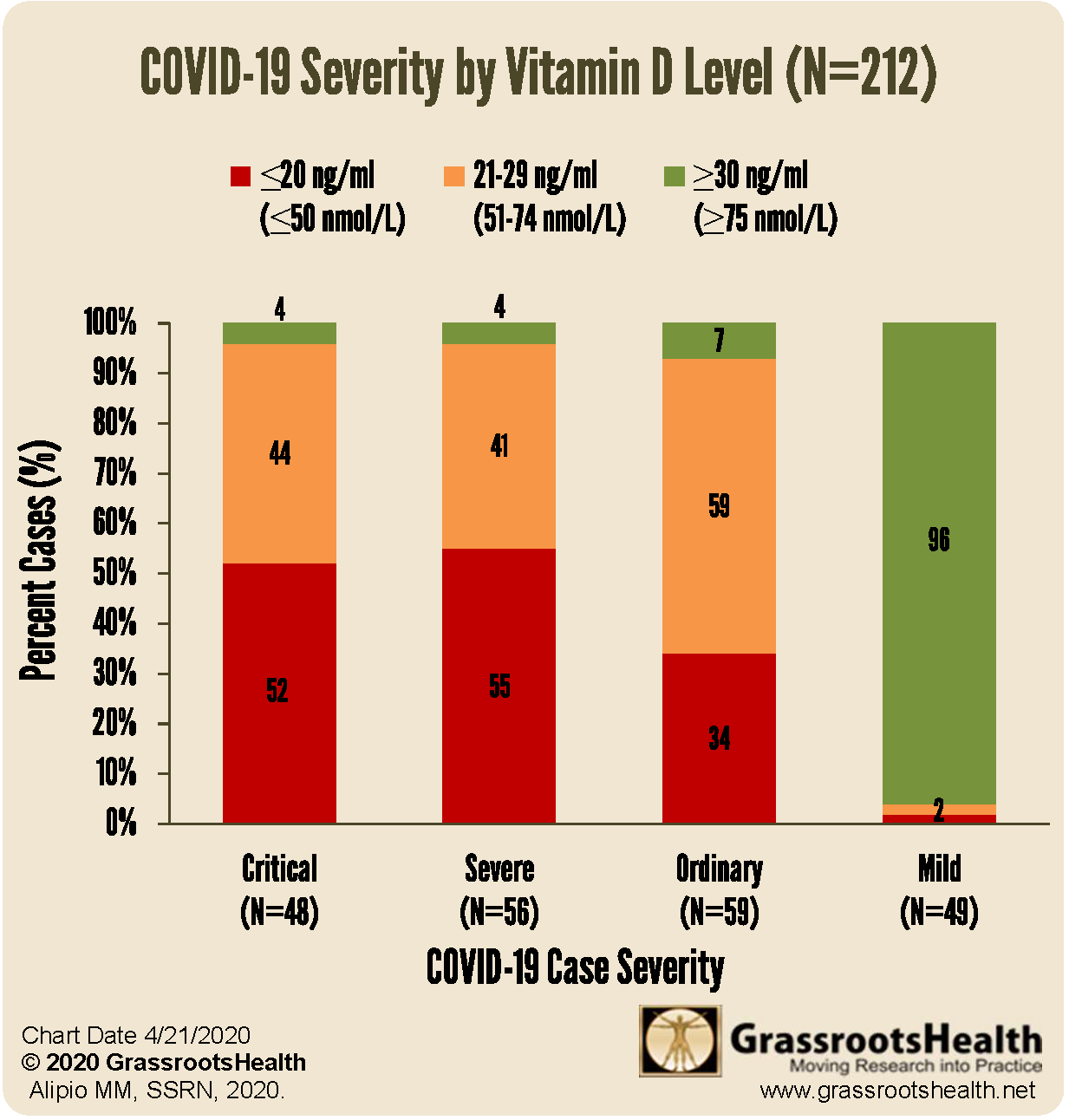
We now have direct evidence linking deaths and critical outcomes to COVID-19 related deaths from studies yet to be peer reviewed. This following graph from Grassroots Health summarise the results of a small study from the Philippines [11]. It illustrates that only 4% of those who had vitamin D blood levels above 30ng/ml became critically or severely ill, whilst 96% of those whose levels were below 30ng/ml became critically or severely ill. 96% of mild cases were found to have sufficient, >30ng/ml, blood levels of vitamin D. The researchers did not look at optimal levels above 40ng/ml.
 Indonesian research published more recently and updated on 6 May using data from 780 patients who had tested positive for COVID-19 [71] have confirmed these findings. While only 4% of patients with sufficient levels of vitamin D (>30ng/ml (OH)D) died, the mortality rate was 87.8% and 98.9% in the insufficient (20-30ng/ml) and deficient (<20ng/ml) groups, respectively. After adjusting for confounding factors such as age, co-morbidities and sex, the researchers still found that death was 7.63 times as likely in the insufficient group and 10.12 times as likely in the deficient group.
Indonesian research published more recently and updated on 6 May using data from 780 patients who had tested positive for COVID-19 [71] have confirmed these findings. While only 4% of patients with sufficient levels of vitamin D (>30ng/ml (OH)D) died, the mortality rate was 87.8% and 98.9% in the insufficient (20-30ng/ml) and deficient (<20ng/ml) groups, respectively. After adjusting for confounding factors such as age, co-morbidities and sex, the researchers still found that death was 7.63 times as likely in the insufficient group and 10.12 times as likely in the deficient group.
These are highly interesting findings, and if taken in isolation, the question of whether deficiency led to the poor outcomes or whether the poor outcomes depleted serum vitamin D might well be a legitimate one. It is acknowledged that vitamin D may well become depleted during the course of an infection, perhaps more so in the critically ill. However, to argue for the second alternative scenario would require a narrowly focused dismissal of all the other evidence and facts noted in this report.
On the contrary, when taken together with the large body of interventional studies on respiratory infections, the biochemical mechanisms outlined above which illustrate strong biological plausibility, the correlations between countries discussed above, and the knowledge that high risk groups are almost certain to be vitamin D deficient to begin with, the results of these latest studies point strongly to the conclusion that there is a compelling case implicating vitamin D deficiency in COVID-19 outcomes and deaths.
7. Cautions Regarding Trial Design and Explanation of Negative Findings
If larger scale UK specific interventional studies are conducted to confirm the above findings there is an important word of caution. This is that because, quite unlike other nutrient interventional studies where one group receives a supplement and the control group receives a placebo, it is quite possible for the control group to end up with higher vitamin D levels because of sun exposure, rendering the comparisons between the groups and therefore the test results meaningless. Hence, it is critical that regular blood tests of serum 25(OH)D levels are carried out at appropriate intervals during the test period to ensure that the test group does in fact have significantly higher blood levels of vitamin D than the controls. Dosing of the test group should also be daily (see below) with D3, not D2 [63,64].
Vitamin D increases calcium absorption: therefore, calcium supplements should not be taken in excess at the same time. If there is a concern about osteoporosis, it is of note that countries with the highest calcium intakes have some of the highest rates of osteoporosis. What is required to build bone density is often not more calcium, of which most of us obtain adequate amounts in our diets, but sufficient vitamin D3, K2 and magnesium, all of which many are far more likely to be deficient in.
Most studies that found vitamin D unhelpful used either; far too low a dose; a weekly or monthly dose rather than a daily dose; D2 not D3; a single extraordinarily high dose (eg.100,000 IU or greater bolus dose); or did not ensure that blood levels of vitamin D were much higher than those of the controls or sufficiently high i.e. above 40ng/ml [53,54]. In others, lack of other limiting vitamin and minerals such as magnesium and K2 may also have been a factor [58-60] and therefore the status of these also needs be determined in any rigorous vitamin D trial.
Magnesium is required for the conversion of vitamin D to its active form, calcitriol [59,60]. As a high proportion of the UK population is magnesium deficient, supplementation with magnesium may also be required to gain the benefits of optimal vitamin D3. In addition, vitamin A and D work together and the levels of vitamin A must also be adequate. Vitamin K2 may also need to be supplemented in most people as it has been shown to work with vitamin D to keep calcium out of arteries and hence reduce coronary heart disease (,as well as to pull calcium into bones) [58] and this is a nutrient that many people are deficient in.
8. Daily Requirements and Concerns Regarding Toxicity and Sun Exposure
It is preferable to obtain vitamin D from sensible sun exposure than supplements as the half-life in the body is 2-3 weeks for supplements but double that for vitamin D produced in the skin. Vitamin D produced by the action of sunlight is stored in subcutaneous fat cells under the skin which release it gradually [73 p147].
In the UK, average blood levels are just 19 ng/ml, under half the minimum optimal level. Whilst the daily dose suggested by Public Health England of 400 IU/10 mcg may be adequate to ward off the effects of severe deficiency which results in rickets, it has been demonstrated to be far too low to achieve blood levels of 40-60 ng/ml 25(OH) vitamin D, consistent with an optimal balanced immune function and the prevention of many other diseases. Even 2,000 IU daily has been shown to only achieve serum levels of 30ng/ml [80].
Studies show that almost 6,000-10,000 IU per day is required to maintain optimal blood levels of 40ng/ml levels [5,80]. This can come from a combination of food, light, and supplements. However, as winter UVB light in the UK is not sufficiently strong to make vitamin D, 6,000-10,000 IU is required from food and supplements from September to April if optimal levels are to be maintained over the winter. In practical terms this means supplementing a significant proportion of this as very little can be derived from the diet. The darker the skin, the higher the amounts needed from supplementation in summer. Requirements depend on body mass and the obese may require double or triple the average amounts.
In summer, at UK latitudes, white skinned individuals following the recommended sunlight exposure of 15 minutes 3 times a week and wearing shorts and t-shirt were found to achieve blood levels of vitamin D, 25 (OH) D of less than 20 ng/ml, only enough vitamin D to prevent rickets [88]. They did not achieve optimal (40-60ng/ml) or even sufficient (30ng/ml) levels. Therefore, even the white skinned population needs to top up with supplements throughout the summer unless they achieve especially good sun exposure. Even in summer the UVB rays are not strong enough to make much vitamin D before around 10 am or after around 3pm [17].
A white-skinned individual in a bathing suit can make 20,000 IU in 20-25 minutes of sun exposure in high summer [17] at UK latitudes. Synthesis is self-limiting: continued sun exposure does not result in continued synthesis beyond around 25,000 IU. The amount of UVB required to achieve this, 54mJ/cm2 , is termed 1 minimal erythemal dose or MED. It is extremely difficult for a dark-skinned individual to achieve any appreciable increase in their vitamin D levels from sun exposure in the UK. As an illustration, a fair white-skinned person exposed to 1 MED can achieve a 50-fold increase in serum vitamin D, but a very dark-skinned person of the same age shows no significant increase after the same exposure. In contrast they would need 5-10 times 1 MED merely to make 60% of the same level [17]. As around 90% of our requirements need to come from sun exposure or supplements (,given food can only provide a small fraction of it) [17], it is clear that BAME populations in the UK are far more reliant on supplements to meet their vitamin D needs. Further, it is clear that with only 400IU per day they will not achieve even a sufficient level.
Given the body’s capacity to make the equivalent of up to 25,000 IU per day, suggestions that levels above 2,000-4,000 IU may be toxic are unfounded. Daily doses of 10,000 IU [8] and even 30,000 IU [65] have been firmly demonstrated to be perfectly safe [65,61,62,66]. Toxicity is extremely rare and a review of 15 studies show that toxicity is not seen until blood levels reach above 200ng/ml [65]. To put that into context, lifeguards are known to maintain blood levels of above 100ng/ml without ill effects [9,17,61,62]. The European Food Safety Panel’s Statement of Scientific Opinion acknowledges that no adverse effects have been observed at 10,000 IU or 250 mcg per day [8]. People with impaired kidney function should be closely monitored and remain under the guidance of a medical professional.
Concerns about cancer dangers from sun exposure may be misconstrued. Almost 90% of skin cancer deaths are caused by melanoma. Although there is an association between excessive sun exposure and non-melanoma skin cancers, and excessive sun exposure or burning should be avoided, there is no credible scientific evidence that moderate sun exposure can cause melanoma[17,77,61,62].. Working indoors and lack of vitamin D is associated with a significantly increased risk of melanoma while occupational sun exposure is associated with reduced risk of melanoma [77]. Melanoma is more often found in areas which are not exposed to the sun [17,62]. There are other risk factors for melanoma such as being obese, high alcohol consumption and being sunburnt when young, but sun exposure in the present which is no more than moderate is not a risk factor. Ironically, 83% of Australian dermatologists were found to be vitamin D deficient [29].
The dangers and extensive ill effects on health of a lack of moderate sun exposure and consequential lack of vitamin D, appears to, by far, outweigh any health benefit from avoiding it. It is possible that sun exposure may have other unknown positive effects on the human body besides making vitamin D. A Swedish 20 year follow up study of almost 30,000 women found that avoidance of sun exposure is a risk factor for death of similar magnitude as smoking! [89].
Conclusion
It is clear that the existing medical literature on the crucial role of vitamin D in fighting respiratory infections and controlling overactivity of the immune system, is abundant and highly persuasive. There is new evidence that maintaining high vitamin D serum levels is highly protective against COVID-19. Furthermore, studies have already established deficiency in a higher proportion of the UK population than in other European countries or the US. The UK population has remained largely vitamin D “deficient” and “insufficient” with average blood levels of only 19ng/ml, far lower than levels of 40-60ng/ml shown to support good health and resistance to infection.
Whilst the daily dose suggested by Public Health England of 400 IU/10 mcg may be adequate to ward off the effects of severe deficiency which results in rickets, it is far too low to achieve blood levels of 40-60 ng/ml 25(OH) vitamin D consistent with optimal immune function. Studies show that 6,000 to 10,000 IU or up to 250mcg per day, up to 20 times the current recommendation is required from a combination of sources, to maintain blood levels of 40ng/ml levels [5,80].
It is recommended that GP testing of blood levels of vitamin D and high levels supplementation is implemented immediately for all in BAME, the obese and other high-risk categories. As a cost-effective intervention with little to lose, low risk of toxicity (Section 8 ) and much to be gained, if efforts are made to raise vitamin D levels, it is likely that the UK will see a lower death rate going forward from COVID-19 and bolster itself against any influenza epidemics or other future pandemics.
Large-scale testing of vitamin D status of COVID-19 patients in UK hospitals and interventional studies may be desirable to gain UK specific information to further inform supplementation guidelines and policy. To avoid a false negative result from trials however, it is critical that for reasons discussed in Section 7, dosing is daily, blood levels of vitamin D are measured at appropriate intervals and the target group’s level is raised to at least 40ng/ml.
All-cause mortality has also been shown in a number of studies to be inversely related to serum vitamin D levels [55-57,89]. Many conditions associated with vitamin D deficiency have already been mentioned but that list is by no means exhaustive. Vitamin D deficiency in pregnancy not only has implications for the mother’s own health but will also cause the unborn child to be epigenetically “imprinted” to be susceptible to chronic disease in later life. A panel of prominent researchers in the field attempted to evaluate the total cost of vitamin D deficiency in western Europe, publishing a paper in 2009 which concluded that raising the population’s vitamin D levels to 40ng/ml could result in savings on a conservative basis, of €187 billion [74]. This translates into an estimated potential saving of around £23 billion per annum for the UK economy*, part of which will relate to direct healthcare costs and the rest to indirect costs. This is a significant sum and represents 16.7% of the current Health and Social Care Budget of £140 billion.
Given the likely deeply negative impact of the current crisis on the economy in the medium term, it is imperative that the UK finds effective ways to manage health costs going forward. Vitamin D testing of serum 25(OH)D added to GP’s routine tests with incentives for achieving levels of at least 40ng/ml in patients has the potential to significantly reduce future healthcare costs.
* based on savings being the same percentage, 16.7%, of total health costs as calculated in the paper.
References can be viewed below (please click the ‘pop out’ box in the top right corner):
 Loading...
Loading...
About the author
Eshani has been passionate about science from a very early age. Following a degree in Biochemistry with an option in immunology at Bristol University, Eshani changed direction and qualified as a Chartered Accountant with KPMG, specialising in corporate taxation. In 1998 Eshani left her accountancy career in order to devote time to carrying out research to help alleviate the serious health issues that one of her close family members developed.
As a member of the BAME community with family members who potentially have a high risk from a COVID-19 infection, she was highly motivated to examine existing and recent research supporting the role of vitamin D deficiency in COVID-19 outcomes.
With many thanks to Eshani King for this detailed article. If you have any questions regarding the health topics that have been raised, please don’t hesitate to get in touch with Amanda via telephone; 01684 310099 or e-mail amanda@cytoplan.co.uk
Last updated on 8th March 2021 by cytoffice



What a wonderful article. It brings together so many bits and pieces of information floating about on news sites, etc. The monitoring of the vitamin D level should be a regular procedure for all patients visiting the doctor for a check up or control visit.
Thank you,
Ted Green
This is an excellent article! Having always supplemented Vit D because I work in a windowless office it’s reassuring to know.
Thank you.
Fascinating! I’ve seen what vitamin D has done to normalise the function of my parathyroid glands and in doing so has improved my bone density as shown by DEXA scan.
This is the best study on the effects of lack of vitamin D that I have seen even though I keep up with as many studies and articles as possible. It is particularly of interest to me as I’m 68 and very slightly built with sinus problems probably linked with chronic migraine. My vitamin D was very low and I have been supplementing with 1000 mg of vitamin D for 6 months. It made me realise that 2000 mg of vitamin D would not be harmful, and infact beneficicial, and also to consider K2 and daily magnesium as an adjunct. I worked in an homeopathic clinic for several years and natural health ideas are important for prevention of viruses.
This is such a great article which pulls together, in one place, much of the research on the importance of Vitamin D. Thank you so much for this!
A very good blog with much data and well researched. Confirms my view that Vitamin D is crucial for protection against many chronic diseases. I think more should be made of the synergistic role of Magnesium and K2. You should not be taking 5000-10,000 IU of D3 without K2. I also have read Zinc and Selenium are important in boosting immune system. Vitamin D on its own is not a magic bullet and the above added supplements, micro-nutrients and a good plant based diet with limited sugar and gluten, exercise and good sleeping patterns are all key to staying fit and healthy. Perhaps a sentence to that effect would be useful!!
Having Vit D blood levels measured is also critical to ensure you are achieving a desired level in your system. Minimum of 2x a year and if GP wont do it you can order on line and get a blood prick test for £29 and done in an NHS lab in Birmingham.
Hopefully our UK Drs will finally start to take Vitamins and nutrition seriously rather than their continued obsession with pharmaceutical drugs as their only prescription.
Thanks for the work on this critical topic. Vitamin C could be another good Blog especially IV for treating Covid and Cancer.
Excellent post. Have shared.
Again another excellent article.
We need this in the newspapers! Pronto.
I have been on a D3 and K2 spray bought in Oz plus a good multi from Cytoplan extra mg and vit c, I also added glutathione at the beginning of outbreak and only suffered very mild symptoms. My daughter and family were staying with us and were tested positive on their return to Oz.
Very helpful to know how much more we can safely take.
Many appreciative thanks
Thank you for this revealing and insightful article.
Thank you for this comprehensive research regarding Vitamin D3 and the Immune System, BAME and Covid-19 Pathogen.
As a young adult I was diagnosed with Asthma, after a traumatic event. During childhood and adolescence I constantly had one chest infection after another.
Culminating with a Victorian disease QUINSY, which was almost fatal and soon after a tonsillectomy.
In 2008 I attended the Mandala Yoga Ashram in Carmarthenshire, where I was introduced to specific breathing techniques. I practiced these skills,now key tools, for many moons.
After suffering from Asthma for thirty two years and many yoga courses later, including travelling to India, Bihar School of Yoga and Holland, stillnessinyoga, I have optimum health for my age.
Guess what! A third lot of blood tests four years ago, revealed my lack of vitaminD3.
FULL OF GRATITUDE
Impressive article. I was aware of some of the benefits of vitamin D, but did not realise how potentially crucial it is to the health of this country, particularly in fighting Covid 19. I will increase my dose of sunshine and supplements. I hope the powers that be see fit to respond to studies such as this and find ways to ensure the population at large benefits from the findings.
Then great use of ghee by indian subcontinent cultures and widespread type B diabetes worth considering as well no doubt due to the love of sugar laden foods
I don’t agree vitamin D is very important but it does not explain why men are more prone to C19 than women and children it seems to me to skip over a lot of the questions and if it was down to lack of vitamin D countries who get a lot of sun would not be affected with a lack of vitamin D I think this article is a case of making the foot fit the boot
Hi Dean,
Vitamin D is a nutrient that has been demonstrated to play a significant role in protection from Covid-19 and also how badly it affects individuals. But it by no means stands alone. We have written several other blogs on the many other susceptibility factors which you might find of interest. Men appear to be worse affected because major genes coding for innate immunity are located on the X-chromosome. Women have two copies and men only one. The interaction of these genes together with their generally higher vitamin D status as well as hormonal factors are thought to result in women being less adversely affected by COVID-19 and by all infections generally than men.
Thanks,
Amanda
There is also emerging evidence that men have more ACE2 receptors than women. ACE 2 receptors are the binding and entry sites for Covid 19.
Just because a country has a lot of sunshine doesn’t mean that the population has adequate exposure to the sun. The “sunsmart” message to cover up and use sunscreen means that too many people have inadequate sun exposure. As mentioned in the article, in some cultures people (especially women) cover their bodies and faces – think Iran at the beginning of the pandemic and the high rate of infection and mortality there.
What a brilliant piece of work. I have supplemented with VitD3 &VitK (from Cytoplan) for many years. I also take Magnesium (amongst other things). I very rarely get colds – and if I do, they generally last only 2-3 days. I started to supplement after being diagnosed with cancer, as I read that Vit D3 underpins the immune system and thus helps your body fight many infections and cancers etc.
When reports first surfaced regarding the BAME communities, I wondered about possible Vit D deficiencies, due to darker skins and our less fierce sunshine. I had read in the past that there was a higher tendency for the BAME communities to develop rickets – again from a lack of Vitamin D, so it seemed highly possible that the lack was a factor in their responses to Covid-19. What I had not thought about was that a lack of Vit D in the elderly – but again, it really makes sense that they are often also deficient in Vit D.
Congratulations to the author for giving us all the information so clearly. I hope that the report has been forwarded to the Health Secretary.
Excellent article, congratulations.
Note of caution; high dose Vit D supplementation is safe (ibid) but may disrupt the fine balance with other essential nutrients, in particular Vit K2, which is also frequently low.
I have a VDR SNP which I support with D3 and K2 supplement.
Hi Robin,
Yes, I concur that ideally supplementation should be individualised and blood levels of Vitamin D and other nutrients monitored, if one is taking high doses of any nutrient over the longer term. Genetics are particularly important for the level of Vitamin D (and K2) an individual might need and those with the VDR SNP often need significantly more.
Thanks,
Amanda
Excellent article which pulls together so many areas of research into covid 19. Will share with all my nutritional and naturopathic colleagues.
An excellent article. I’d like to have a copy of the references.
Hi Mike – we will email these to you today.
An excellent piece of work, Eshani, which I will share as widely as possible!
Many thanks to Eshani King for this interesting and well-researched article. Any more such articles by Eshani would be very welcome!
Brilliant article – better than any I’ve read on vitamin D and COVID-19 in the last three months, and I’ve read a fair few! The best explanation I’ve read about how vitamin D fits in with the ACE2 receptor issue. Thank you!
Very interesting article, thank you for it!
Excellent. Informative and fascinating.
Always look forward to reading your posts , you’ve excelled yourselves with this well thought out and researched article . Keep up the great work your doing in the world
Excellent post! Is there an easy way of working out how much magnesium and K2 one should be taking to ensure most effective use of supplementation by Vitamin D?
Hi Rose,
A good daily supplemental intake for the general population of these nutrients is 60-100ug K2 and 80mg-200mg elemental magnesium. The latter will equivalent to 1-2x 500mg magnesium citrate.
500mg is the compound weight and the elemental is 80mg. But, as with all supplementation, it is best to individualise and take the level needed after a review of, diet, lifestyle, genetics and presenting problems. We offer a free health questionnaire if that’s of interest – here.
Thanks,
Amanda
Many thanks, Amanda – I’ve used your valuable questionnaire service before. I think (with 2 x Cytoplan Bone Support + 1 x CoQ10 Multi per day) I should be OK on the K2 front … Looks like I need to up my Magnesium though!
Agree need to get regular tests and then adjust your supplements. I have found as I get to close on 70 have to increase dosage to get a decent blood level.
Excellent and very informative. I found it extremely helpful. I have heard so much about the value of Vitamin D but didn’t actually realise how very important it is for my health and well being. Thank you Eshani
Re the premise – virus degrade in heat : I have heard this assumption many times in relation to the seasonal ‘flu, however, with regard to Covid 19 – can we make this assumption as Brazil appears to be doing rather badly ? This is a sunny country and probably has a higher Vit D status in the population, albeit perhaps not enough. Since I am of the opinion ( from research I have mostly read from USA) Covid 19 might be a Coronavirus, but there the similarity ceases as it was Man Made. I therefore think it dangerous to draw any comparisons with how the virus might behave.
Comments most welcome.
An excellent article on vitamin D. Most comprehensive and thorough. Well done and thank you. I hope the powers that “be” within our health service have access to it. They might catch up a bit quicker before the next onslaught !
Hi Marilyn,
Thank you so much for your comment on the blog, you raise some interesting points. The virus has been fully sequenced and its genes identified – it is continuing to mutate (though nowhere near as rapidly as influenza) but scientists are keeping track of mutations and much of its behaviour, though not all, is ascertainable. RE: Brazil – the paper does not claim that vitamin D status is the only determinant, only that is a major one. Yes, Brazil has done worse than other sunny countries and there are clearly other factors at play in Brazil but even so, Brazil is nowhere near as badly effected as the UK (or most other European countries) with 272 deaths per million population as reported today, compared to UK’s 642 deaths per million. As Brazil’s population is 213 million as compared to UK’s 67 million, and in any event the number of cases in any country is highly dependent on the extent of testing, it is not meaningful to compare numbers of cases. Deaths per million is a more meaningful comparison although even then, this figure is dependent on how far a country has got in terms of infections spread.
I hope this helps answer your questions. Please let me know if I can be of any further help.
Thanks,
Eshani
Congratulations Eshani! Terrific work, bringing together so many studies into one coherent & understandable article. The poor levels of the UK populations vitamin D levels could be addressed systematically, simply & cost effectively by governmental intervention prior to the Winter. Failing that, of course, taking personal responsibility is essential. I will be sharing the article & ensuring that my whole family & friend network know the key takeaways.
Muchas gracias.
Muchas gracias.
excellent article.I get respiratory difficulties most winters and now realise that spending a lot of time outdoors hardly helps when you live in a thickly clouded hilltop and cover up against the rain.I will increase my vit d to try surviving this winter. Nutrition needs to be more strongly promoted in medical training..It would be good to print this out and stick copies on the surgery wall, if permitted
by the Practice.
This was a very good and informative blog, but the arguments would have been made even more effectively if Eshani had addressed the countervaling views that currently predominate in organisations like the WHO and NICE. NICE’s position may be influenced by the fact that testing serum Vit D levels is a relatively expensive test for the cash strapped NHS. I write this as someone who does believe that the Vit D story is important and currently take Vit D supplements in the hope that I might derive some protection from Covid, but it’s important that articles written in support of Vit D can’t be accused of selection bias in their choice of research references.
Two unrelated points. I would like to see research on the relative rates of morbidity and mortality in men and women in devout Islamic countries where women tend to cover up more than men. Theoretically women in these countries might tend to have lower Vit D levels and therefore there might be a difference in the ratio of men to women who are severely affected by Covid. The confounding factor would of course be that women in those communities mix less and therefore their infection rates would be likely to be lower.
Secondly re morbidity in children. We know that infants and preschool children have an incredibly high frequency of mild viral infections. I suspect that short lived cross immunity with other Coronavirus infections may explain why young children so rarely manifest Covid symptoms (as you partly implied) and may also be less infectious when they carry the virus. I would love to see research into whether professions which have frequent contact with large numbers of small children, and therefore possible frequent exposure to trivial coronavirus infections, (e.g. primary school teachers, paediatric doctors and nurses) show evidence of protection from Covid.
Hi Nick – thank you for your comment on our blog. Eshani has quite a detailed response to your questions so we will be emailing this over to you in due course.
I read the evidence of the efficacy of Vitamin D3 in prevention of various conditions in March when Covid first hit the headlines. I have been taking a Daily does of 5000 IU of Vitamin D3 daily since then. It costs a few pennies a day. I have had no colds, flu or any other respiratory ailments. I will be taking it for life as I am so convinced of its properties. I lived in Southern Spain for several years and noticed the difference there. I am now back in NE England, so continuing to take it is even more important, my family are all taking it too.
That’s great to hear, Andrew 🙂
Thanks for such a brilliant article; so clearly explained.
Very interesting, informative and well researched article
What an absolutely brilliant article. I have been taking high strength Vit D for some years now, and recently with K2. My arthritis has really improved, and I can’t remember the last time I had a cold!!
This article should be put in front of every MP & decision maker in this country and around the world, but unfortunately it won’t bring in the revenue .
Thank you. This is not only a brilliant article but the responses add another layer of information. I’m posting it to many people in the hope that they will take the time to read it.
Given the evidence, it is hard to see why vitamin D has not been a public health objective in the UK, and in particular why virtually nothing has been heard over the past year of its potential role in combating Covid-19.
In our household we have been taking 4,000IU per day; having read this article we shall be taking 8,000IU per day from now on.