A complex topic that can be baffling and spark differing opinions, methylation is critical to health as it’s at the heart of so many of our body’s key processes. Annie Edwards demystifies methylation and explains how you can use it to support the health of your clients.
Why methylation matters
Methylation is a key cellular process that takes place continuously, regulating multiple functions in the body every second. It works by adding a methyl group to a substrate, which can activate or deactivate it, acting like a molecular “currency” that enables countless physiological “transactions” throughout the body. Methylation also plays an important role in epigenetic regulation, switching genes on or (more commonly) off, enabling us to modify our genetic code ‘on the fly’, and downregulate gene expression that might be deleterious to health. We simply can’t live without methylation!
This whole process is dependent on two key biological cycles – the folate and methionine cycles, which produce 5-methyltetrahydrofolate (5-MTHF) and S-adenosylmethionine (SAMe), the body’s primary methyl donor. Effective methylation relies on an adequate supply of methyl donors, nutrients involved in single-carbon metabolism that can donate a methyl group and alter the activity of other compounds. These include folate (particularly in its active 5-MTHF form), vitamins B2, B6, and B12, zinc, choline, and betaine trimethylglycine (TMG). Different folate derivatives support DNA and cell replication and repair, while 5-MTHF specifically supports neurotransmitter and hormone synthesis as well as being the key substrate for SAMe, which donates methyl groups via methyltransferase enzymes to support a wide range of essential methylation-dependent processes.[1][2][3][4]
As methylation underpins so many important body processes, it can easily be disrupted by our environment, including nutrient intake, stress, sleep, and gut health. Dysregulation has been linked to various health conditions such as Alzheimer’s disease,[5] depression,[6] anxiety,[7] osteoporosis,[8] and cardiovascular disease.5 There are also some very common genetic polymorphisms (SNPs) that can affect the methylation process at different levels. So, our ability to methylate can be very individual and often requires tailored support.
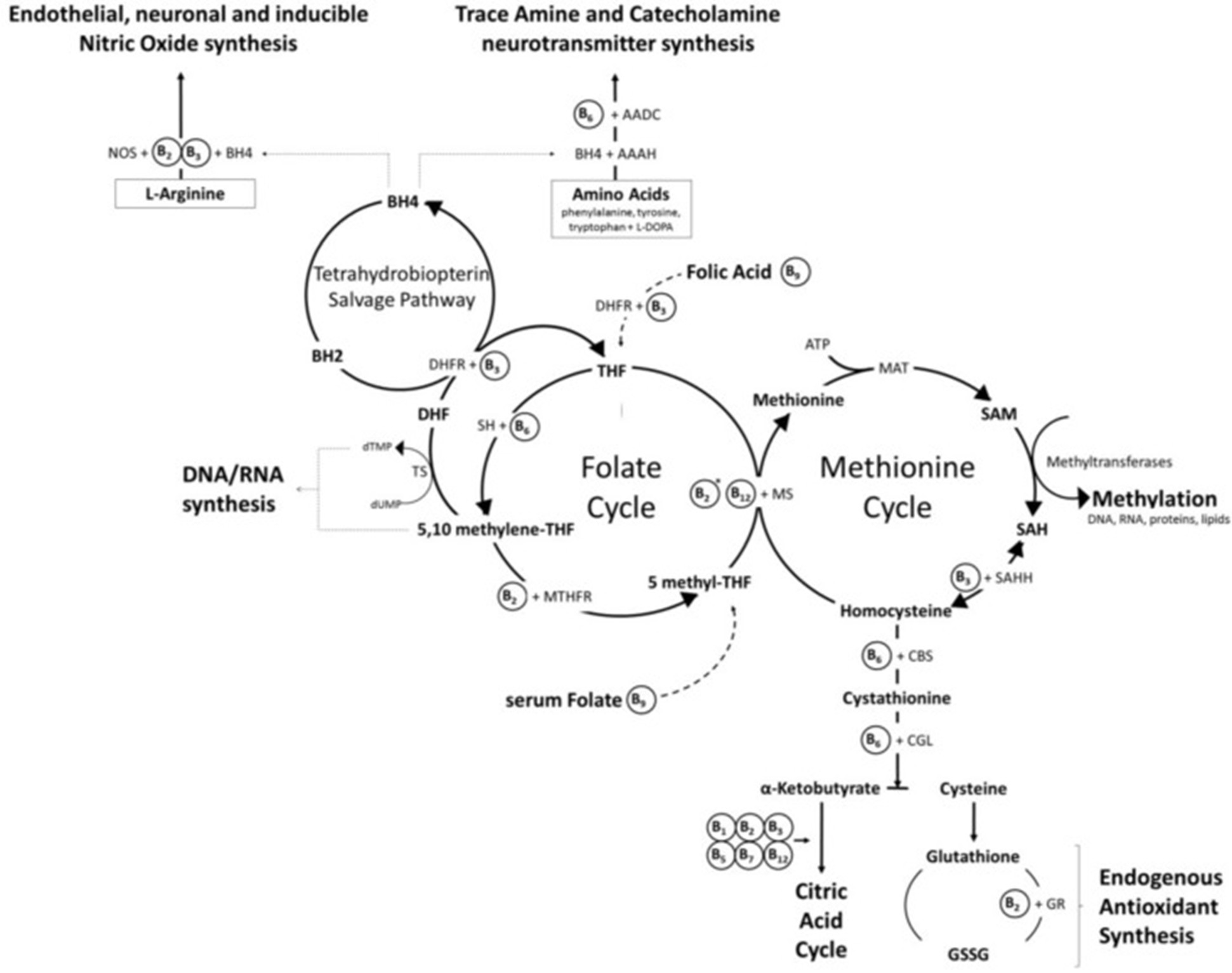
Diagram[3]
Why do we need to methylate?
Methylation significantly influences every aspect of health. It plays a vital role in all biological systems, including:
The nervous system: Methylation plays an essential role in nervous system health by supporting the synthesis of key neurotransmitters and neural compounds, including dopamine, serotonin, adrenaline, noradrenaline, melatonin, myelin, and acetylcholine, all of which are vital for mood regulation, stress response, sleep, nerve signalling, and overall brain function.
Cardiometabolic health: it supports the synthesis of key molecules such as CoQ10 and carnitine, essential for cellular energy production and fat metabolism. It also plays a role in the production of nitric oxide, supporting healthy circulation, and in the metabolism of homocysteine, helping to maintain cardiovascular health and metabolic balance.
Endocrine and energy systems: it supports the synthesis of key compounds, including tyrosine, CoQ10, carnitine, creatine, adenosine, and NADPH. These are essential for hormone production, cellular energy generation, mitochondrial function, and redox balance, helping to regulate energy levels, metabolic efficiency, and overall endocrine health.
Sex hormones, fertility, and pregnancy: it plays an important role in oestrogen metabolism, helping to support hormonal balance. It is also involved in the development of healthy sperm and ova, and supports normal foetal growth and development, making it a critical process for reproductive health across all life stages.
Digestion and liver health: it supports the synthesis of phosphatidylcholine, a key component of bile that helps with fat digestion and supports liver detoxification.
Detoxification: it is a key Phase II pathway, supporting the processing and clearance of hormones, catecholamines, and heavy metals. It also contributes to glutathione synthesis and sulphation processes, which help neutralise and eliminate toxins. It further plays a role in histamine metabolism and supports balanced immune and inflammatory responses.
Structural health: it supports DNA synthesis, cell differentiation, and gene expression, while also helping protect DNA through epigenetic mechanisms such as histone modification. It contributes to the production of phosphatidylcholine, an essential component of cell membranes, and plays a role in bone formation.
Immune health: methylation supports the production and function of immune cells, helping them develop, recognise antigens, and respond effectively to threats. It also plays a role in antiviral defence and tumour surveillance, contributing to balanced and resilient immune function.
What is homocysteine?
An important function of methylation is its ability to metabolise the amino acid homocysteine. Homocysteine is produced during the conversion of methionine to SAMe in the methionine cycle and relies on vitamin-derived cofactors such as vitamins B6, B12, and folate. While we need some homocysteine to make proteins in the body, in elevated amounts, it becomes an inflammatory substance that can promote blood clot formation and negatively affect endothelial and, therefore, cardiovascular health. During methylation, homocysteine can either be remethylated back to methionine by the acceptance of a methyl group or be converted into cysteine via the trans-sulphuration pathway, which is important in the production of the antioxidant glutathione. Homocysteine can be raised by lifestyle and health factors, including alcohol consumption, smoking, kidney disease, physical inactivity, inflammatory conditions, hypothyroidism, and use of certain medications, such as proton pump inhibitors (PPIs).
What can influence methylation?
Methylation can be highly responsive to external and internal changes, including environmental toxins, poor nutrient intake, stress levels, and genetic SNPs.1
Nutrition
A diet high in processed foods, excess sugar, and alcohol, along with smoking, can impair nutrient absorption and disrupt methylation by reducing the availability of key nutrients and increasing their breakdown in the body. Low intakes of nutrient cofactors such as choline, folate, B2, B6, and B12, and the minerals zinc, magnesium, and iron can disrupt methylation. In order for the methylation cycle to work effectively, nutrients must be converted into their active form in the body. For example, B12 as cyanocobalamin must be converted to methylcobalamin, if it is not directly supplemented as such, whilst folic acid (found in fortified foods and most high street supplements) must be converted into the 5MTHF form.[9] [10] This latter form of folate is thought to be more effective than standard folic acid.
Lifestyle
Chronic stress, hormonal imbalances, poor sleep, and exposure to environmental or chemical toxins, including heavy metals and endocrine disruptors, can all disrupt methylation. In addition, factors such as sub-optimal digestion, use of PPIs, and other medications may impair nutrient absorption, leading to reduced availability of essential methylation cofactors.
Gut health, as always, is another important consideration concerning both methylation and energy production. Dysbiosis, low stomach acid production, and low levels of B6 and zinc (needed for stomach acid production) can impair nutrient absorption, while reduced protein digestion can also directly affect methionine levels.
Genetic SNPs, including MTHFR
Certain polymorphisms can impact methylation and subsequent energy production. For example, as mentioned, methylation needs folate in its active form 5-MTHF, which is usually converted (although poorly) from folic acid. The MTHFR gene variant reduces the efficiency of the normal conversion of folic acid to the bioactive folate, an important methyl donor. SNPs in MTHFR gene are common (two key variants – C677T and A1298C), with approximately 70% of people carrying at least one of these SNPs. Both variants are associated with higher homocysteine levels, particularly when folate and vitamin B12 status are low, although the C677T variant carries the greater risk. Individuals who are homozygous for C677T may experience up to a 70% reduction in enzyme activity, while homozygous A1298C is associated with up to a 40% reduction, and compound heterozygous individuals may have up to a 60% reduction in MTHFR function.[11] In this instance, there is an increased importance of using a form of folate called methylfolate, which bypasses the MTHFR enzyme.
The methionine and folate cycles are linked via an enzyme called 5-methyltetrahydrofolate-homocysteine methyltransferase (MTR). MTR accepts a methyl group from the folate cycle and donates it to B12, which results in methylated B12, ready for use in its role of remethylation of homocysteine and the conversion to both methionine and SAMe. SNPs here can slow this process and interfere with the cycle. B12, once used by MTR, needs to be reactivated via methylation, which is done by the methionine synthase reductase (MTRR) enzyme. This enzyme is specifically impacted by heavy metal toxicity and results in reduced levels of methylated B12, making supporting methylation and reducing heavy metals paramount. Supplementing methylated B12 and zinc as a cofactor can also support MTR.
How can I support methylation?
Supporting methylation is best approached as a two-way process. The first step is to reduce the demands placed on these pathways by addressing the factors that draw heavily on them. This includes lowering stress levels, maintaining an appropriate balance between physical activity, rest, and recovery, and reducing sources of inflammation. Minimising toxic load, from heavy metals, moulds, pesticides, and excessive stimulants, while supporting detoxification pathways, can also ease the burden. In addition, regulating blood glucose and supporting healthy hormone balance help create a more stable internal environment in which methylation can function efficiently.
Nutrition
A nutrient-rich anti-inflammatory diet, rich in an abundance of fruits and vegetables and including good-quality protein, is essential for optimal methylation. It is advised to limit sugar and refined carbohydrates, which contribute to blood sugar dysregulation, alongside limiting alcohol and caffeine, as these can deplete B vitamins and increase stress hormones, which all put additional strain on methylation. Specific foods to focus on include folate rich foods (leafy greens, asparagus, citrus fruits, broccoli, legumes, beetroot, nuts, and seeds), B12 containing foods (mostly animal products or supplement especially if vegan), vitamin B6 foods (wholegrains, nuts and seeds, legumes, meat, and garlic), choline (eggs, meat, shitake mushrooms, legumes, and cruciferous vegetables), zinc and magnesium-rich foods (such as green leafy vegetables, nuts, seeds, and wholegrains). Pre and probiotics – including plenty of fruits and vegetables, as well as fermented foods such as kimchi, kefir, and sauerkraut can support the microbiome and short-chain fatty acid production. Certain bacterial strains, including Lactobacillus and Bifidobacterium, support the production of various nutrients, including folate.[12]
Supplementation
Alongside reducing demand, it’s important to provide the right nutritional support. This typically includes folate in its active methylfolate form and vitamin B12 as methyl- or adenosylcobalamin. Folic acid is often used in supplements, but requires multiple conversion steps to convert to 5-MTHF. However, this process is often suboptimal, especially with SNPs that affect DHFR or MTHFR enzymes, for example, and may lead to a build-up of unmetabolised folic acid, which acts as a block to methylation. This leads practitioners to opt for the preferred form of 5-MTHF.[13] Some practitioners report that the methyl forms can be overstimulating as they are so effective, but there is no hard evidence of negative impacts, and methylfolate is always the preferred form, whether there are SNPs or not. In susceptible people, the level of folate may need to be reduced to suit and, of course, given in context of wider support, by ensuring their stress levels are helped, for example. Other key cofactors such as zinc, vitamins B2, B3, B6, and magnesium are also useful.
A well-formulated methylated multivitamin or B-complex, alongside other foundational “pillars” like essential fatty acids, probiotics, and antioxidants, can help provide broader system support. Opt for one with 400µg of methylfolate and 400µg of methyl B12 for optimum support, (such as Advanced Multi) or gentler, but effective, support such as CoQ10 Multi, Women’s Wholefood Multi, Menopause Multi, or Men’s Multi. Some people may require additional methylation support or higher levels of folate or B12 to suit their needs, depending on SNPs or their insufficiencies. Methylfolate provides 400µg of methylfolate in a single capsule, or Vitamin B12 Spray can be used to add B12 in 100µg increments. For high dose support, Methyl Factors offers optimum doses of a range of methyl donors, including TMG.
Supplementing methylation – research highlights
Disruptions in folate metabolism are associated with a range of mental health conditions, including depression, bipolar disorder, schizophrenia, autism spectrum disorder (ASD), and ADHD. Supplementation with 5-MTHF has been linked to clinical improvements in symptoms.[14]
Supplementation with 600µg of methylfolate improves fertility in couples where at least one partner carried MTHFR gene variants.[15] Further research suggests that women supplemented with 5-MTHF alongside vitamin B12 have a higher likelihood of achieving pregnancy compared to those using folic acid alone.[16]
High-dose methylfolate reduces blood pressure in menopausal women, likely through its effects on biopterin availability and nitric oxide production.[17] Supplementation with methylfolate, methylated vitamin B12, and vitamin B6 in its active P-5-P form has been shown to reduce homocysteine levels by around 30% in individuals with common methylation-related genetic variants, including MTHFR, MTR, and MTRR.[18]
Conclusions
In summary, methylation is a fundamental biological process that underpins healthy cellular function and plays an important role in multiple physiological processes. While the science behind methylation can seem complex, supporting it in practice doesn’t have to be. Because methylation is influenced by both internal and external factors, a holistic approach is key, one that considers diet, targeted supplementation, genetic variations, lifestyle habits, toxic load, sleep quality, and stress levels. Taking this wider view allows for more personalised and effective support, helping to optimise methylation and overall health.
References
[1] Menezo Y, Clement P, Clement A, Elder K. Methylation: An Ineluctable Biochemical and Physiological Process Essential to the Transmission of Life. Int J Mol Sci. 2020;21(23):9311. Published 2020 Dec 7. doi:10.3390/ijms21239311
[2] Pizano, J.M., Williamson, C.B. (2020). Nutritional Influences on Methylation. In: Noland, D., Drisko, J., Wagner, L. (eds) Integrative and Functional Medical Nutrition Therapy. Humana, Cham. https://doi.org/10.1007/978-3-030-30730-1_18
[3] Kennedy DO. B Vitamins and the Brain: Mechanisms, Dose and Efficacy–A Review. Nutrients. 2016 Jan 27;8(2):68. doi: 10.3390/nu8020068. PMID: 26828517; PMCID: PMC4772032.
[4] Wang Z, Zhu W, Xing Y, Jia J, Tang Y. B vitamins and prevention of cognitive decline and incident dementia: a systematic review and meta-analysis. Nutrition Reviews. 2021;80(4). doi:https://doi.org/10.1093/nutrit/nuab057
[5] Hara J, Shankle WR, Barrentine LW, Curole MV. Novel Therapy of Hyperhomocysteinemia in Mild Cognitive Impairment, Alzheimer’s Disease, and Other Dementing Disorders. J Nutr Health Aging. 2016;20(8):825-834. doi:10.1007/s12603-016-0688-z
[6] Jiang W, Xu J, Lu XJ, Sun Y. Association between MTHFR C677T polymorphism and depression: a meta-analysis in the Chinese population. Psychol Health Med. 2016;21(6):675-685. doi:10.1080/13548506.2015.1120327
[7] McCaddon A. Vitamin B12 in neurology and ageing; clinical and genetic aspects. Biochimie. 2013;95(5):1066-1076. doi:10.1016/j.biochi.2012.11.017
[8] Bahtiri E, Islami H, Rexhepi S, Hoxha R. Relationship of homocysteine levels with lumbar spine and femur neck BMD in postmenopausal women. Acta Reumatologica Portuguesa. 2015;40(4):355-362. Accessed December 23, 2024. https://www.researchgate.net/publication/276849846_Relationship_of_homocysteine_levels_with_lumbar_spine_and_femur_neck_BMD_in_postmenopausal_women
[9] Spence JD. Nutrition and Risk of Stroke. Nutrients. 2019 Mar 17;11(3):647. doi: 10.3390/nu11030647. PMID: 30884883; PMCID: PMC6470893.
[10] Menezo Y, Elder K, Clement A, Clement P. Folic Acid, Folinic Acid, 5 Methyl TetraHydroFolate Supplementation for Mutations That Affect Epigenesis through the Folate and One-Carbon Cycles. Biomolecules. 2022;12(2):197. Published 2022 Jan 24. doi:10.3390/biom12020197
[11] Lupi-Herrera, E., et al. (2019). Polymorphisms C677T and A1298C of MTHFR Gene: Homocysteine Levels and Prothrombotic Biomarkers in Coronary and Pulmonary Thromboembolic Disease. Clinical and applied thrombosis/hemostasis: official journal of the International Academy of Clinical and Applied Thrombosis/Hemostasis, 25, 1076029618780344.
[12] Rossi M, Amaretti A, Raimondi S. Folate Production by Probiotic Bacteria. Nutrients. 2011;3(1):118-134. doi:https://doi.org/10.3390/nu3010118
[13] Miraglia N, Agostinetto M, Bianchi D, Valoti E. Enhanced oral bioavailability of a novel folate salt: comparison with folic acid and a calcium folate salt in a pharmacokinetic study in rats. Minerva Ginecol. 2016;68(2):99-105.
[14] Massoni L. (2024) Folic Acid in Neuropsychiatric Disorders. Med Discoveries. 2024; 3(3): 1133
[15] Servy, E. J., et al. (2018). MTHFR isoform carriers. 5-MTHF (5-methyl tetrahydrofolate) vs folic acid: a key to pregnancy outcome: a case series. Journal of assisted reproduction and genetics, 35(8), 1431–1435.
[16] Cirillo, M., et al. (2021). 5-Methyltetrahydrofolate and Vitamin B12 Supplementation Is Associated with Clinical Pregnancy and Live Birth in Women Undergoing Assisted Reproductive Technology. International journal of environmental research and public health, 18(23), 12280.
[17] Cagnacci, A., Cannoletta, M., & Volpe, A. (2009). High-dose short-term folate administration modifies ambulatory blood pressure in postmenopausal women. A placebo-controlled study. European journal of clinical nutrition, 63(10), 1266–1268.
[18] Pokushalov, E., et al. (2024). Effect of Methylfolate, Pyridoxal-5′-Phosphate, and Methylcobalamin Supplementation on Homocysteine and Low-Density Lipoprotein Cholesterol Levels in Patients with Methylenetetrahydrofolate Reductase, Methionine Synthase, and Methionine Synthase Reductase Polymorphisms: A Randomized Controlled Trial. Nutrients, 16(11), 1550.
All of our blogs are written by our team of expert Nutritional Therapists. If you have questions regarding the topics that have been raised, or any other health matters, please do contact them using the details below:
nutrition@cytoplan.co.uk
01684 310099
Last updated on 4th February 2026 by cytoffice


We’d love your comments on this article
It’s easy, just post your questions, comments or feedback below